Paralever, Suspension and Driveshaft factors.
Also see U-joint angles at the bottom.
Paralever Suspension: What is the "Shaft Effect" and how does the Paralever change things?
This is an alternative description of the Paralever principle for those who are put off by the standard technical gobbledygook. First-timers might want to head over to Tony Foale's website motochassis.com for an older version of the excellent article he wrote for the January 2001 MCN. However, Tony's writing is so concise that it leaves some people behind, hence my efforts here. Rather than try to completely describe the design implications as Tony did, I'll be content to illustrate some principles of the system.
Our model will be a motorcycle with the rear wheel locked to the rear drive. This is a realistic example because in operation, it's that link between the wheel and rear drive that transmits the forces involved, so for a given moment in time we can assume that they are a solid unit. After all, removing that link is the same as coasting, and the effects that interest us occur under acceleration and braking (when torque is transmitted from the rear drive to the wheel).
Introduction
The Paralever suspension was introduced on the 1988 R80GS and R100GS, then used on the 4-valve K and Oilhead bikes (except the R1200C). Whereas the original Monolever system had a single pivot at the front of the swingarm, like most motorcycles, the Paralever uses two links to connect the rear drive to the transmission. This is a "Four-bar" linkage, similar to double-wishbone suspension in cars which keeps the rear drive at a contant angle as the suspension moves. In fact, there are several similarities between the two that can be explored.
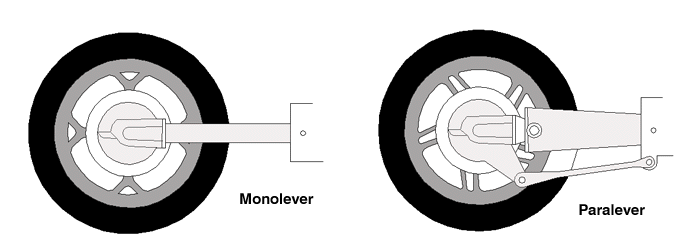
Let's be clear on some terms I'll use here. The "rear drive" is the bevel-gear containing casting that is at the end of the swingarm; it contains the flange that the wheel bolts up to. Whenever I refer to "rotation", I am specifically referring to the rotation, or angular change in position, of the rear drive unit as the suspension moves up and down. The wheel rotates while you ride but that's not important, and the Paralever links rotate around their end points also, but don't let that distract you. We are only concerned with the change in the angle of the rear drive unit (shown as a circle in the wireframe diagrams).
Also, I will mention the forces acting on the rear drive: there is a road force (pushes forward on the tire's contact patch during acceleration) and this is balanced by the inertial force of the bike which pushes backwards at the swingarm pivot. These two forces, since they are not directly opposing each other, create a twisting force or torque in the swingarm/drive/wheel assembly that we're looking at.
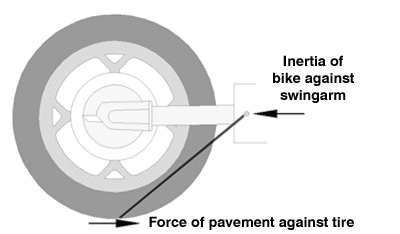
Lastly, it's worth keeping in mind that most of my diagrams portray the two Paralever links as being parallel and of equal length. This isn't exactly correct, but making this assumption clarifies the difference. At the end I'll show how the actual geometry differs.
Operational forces
Refer to the diagrams below while we look at the basic concept: with the Monolever design, you can see that as the rear drive moves vertically, it rotates.
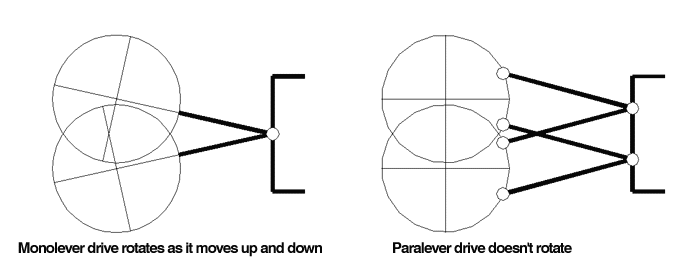
This is a simple result of the fact that the swingarm and rear drive are one unit, hinged at one end: vertical movement will cause rotation, and rotation will cause vertical movement. Think harder about the latter part of that relationship: the torque of the wheel and drive will move the suspension vertically. This torque-induced suspension movement, sometimes called "the shaft effect" or "shaft jacking" is considered to be a drawback to traditional shaft-drive designs. The Paralever drive unit, on the other hand, does not rotate as the suspension moves up and down - it stays at the same angle as the chassis because of the parallel links. Since there is no direct relationship between vertical suspension movement and drive unit rotation, the torque of the wheel does NOT result in suspension movement. This, in a nutshell, is what the Paralever is about. Understand this paragraph and you're almost finished.
If that didn't work, try this: Think about the motion of the contact patch with respect to the bike. Since the Monolever is one solid piece (in our example), every part of that system rotates around the swingarm pivot. As the contact patch pushes forward, it can actually move forward with respect to the rest of the motorcycle by pushing downwards.
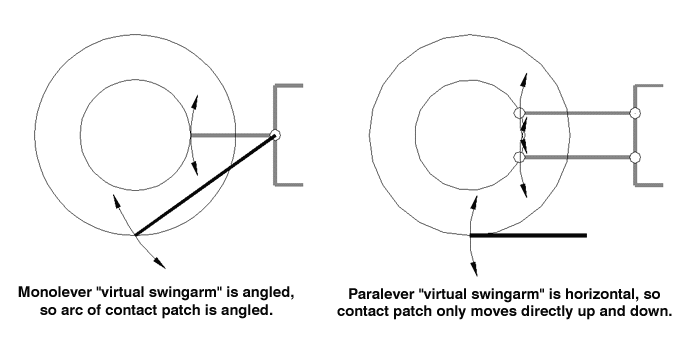
But the Paralever isn't a solid system; the drive is moving without rotating, and each part is moving in the same arc. In fact, the contact patch and rear drive are moving around points that are in thin air! Looking at the arc of motion of the contact patch, you can see that there is no real way for the wheel to try to "scoot underneath" the bike, as the Monolever does. In fact, this "virtual pivot point" is really at the heart of how the Paralever works; BMW has enabled the rear end to pivot around a location where they simply could not put a mechanical pivot.
Here's a automobile example of the same concept. The Monolever is like the "swingarm" rear suspension used on the Corvair. Remember "Unsafe at Any Speed"? The cornering forces tended to raise the rear of that car in exactly the same way, leading to the rollover issue.
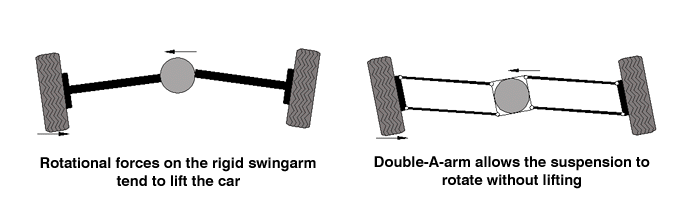
And the Paralever is just like the double A-arm suspension, which is one way the auto makers solved the handling problems inherent to the swingarm. Interestingly enough, another solution to the problem is simply to make very long swingarms, like GM used on the front of their trucks for many years. The torque generated by the cornering force is resisted by a longer lever, resulting in less force at the end of that lever (and less lifting). And the parallel to motorcycles? The R1200C never got the Paralever because BMW considered it unnecessary with the longer Monolever swingarm it uses.
Real-world functionality
Now to be truly accurate, and get to the real nitty-gritty of the design: the geometry of the Paralever isn't an exact parallelogram. As shown below and in the original picture, the forward pivots are closer together, somewhat negating the theoretical benefit. Consider the two extremes: if the two forward pivots were in the same place, forming a triangle out of the rear drive, swingarm and torque arm, all three of those components would rotate around that point exactly like a Monolever. If the linkages form a true paralellogram, like some of my illustrations show, the rear drive won't rotate at all, which would be like having a swingarm of infinite length. By moving the forward pivots closer together (going partway back to being a Monolever) they create the effect of having a much longer swingarm than they really have. The "virtual pivot point" of this system is somewhere around the steering head - a very long swingarm indeed, which resists the shaft effect as mentioned. Additionally, moving the pivot points closer together changes a few things. For one, they gain ground clearance: with the true parallelogram arrangement I described earlier, the forward pivot would just be too low when the suspension compressed over a bump. Primarily, though, they reintroduce enough "shaft effect" to counteract the squat that would otherwise happen under acceleration.
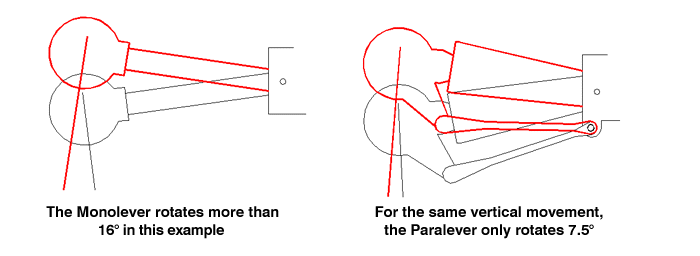
As shown here, the amount of rear drive rotation is cut roughly in half with the Paralever. This means that torque-induced rotation is smaller and will play a smaller role in inducing vertical suspension movement, but there is still enough to counteract the squat that would otherwise happen under acceleration, as weight is transfered to the rear wheel.
(Published in BTS and OTL)
Addendum: I was asked why the shaft effect was something that should be prevented in the first place, and answered:
I'm heartened to note that someone actually read my June article on the Paralever. Carl Wise even read it carefully enough to develop a very good question: "I'm not sure I understand the negative consequences of "shaft jacking." Could you explain this phenomena a bit more to me?"
Right he is. I was so fired up to explain why the Paralever prevented this that I never took time to explain why it should want to. To backtrack a bit, the term "shaft jacking" as I use it refers to the lifting force provided by the suspension under certain conditions described in the article. I used it specifically with the Corvair example, because I first heard the term used that way, but it applies to any similar situation where the fulcrum (usually the inboard end) of a suspension member tries to rise because the outboard end is "tucking under." Think of a pole vaulter. His horizontal movement is translated into vertical movement by a shaft that's angled downwards slightly from his position.
With the car that I depicted, the cornering forces are trying to tuck the wheel under, making the body lift because the swingarm is rigidly attached to the hub. On the bike (you need to rotate your plane of orientation 90 degrees) the wheel is trying to tuck under the bike on acceleration, and the swingarm (again, rigidly attached) tries to lift the bike. By the way, don't limit yourself to thinking about the wheel tucking under and the body lifting, because the reverse happens also. Aggressive riders of older bikes know that they need to stay on the gas in corners, because the chassis will drop down upon deceleration with a corresponding loss of ground clearance.
That last bit cuts to the heart of the negative consequences: the vehicle is no longer riding in its normal attitude (as far as how it sits on its suspension). The accelerating (cornering, etc) forces are pushing parts of the vehicle higher or lower than intended in the suspension range, with two negative effects that I can think of immediately. One is the overall vehicle height; in the case of the car portrayed, the vehicle is riding higher than normal, and is therefore more prone to rollover. The other is the reduced suspension functionality. The suspension travel exists to absorb irregularities in the surface; it should normally stay in the intended region of that travel so as to be ready to absorb whatever bumps or dips the designer anticipated. If the vehicle is pushed towards one extreme of travel by the accelerating forces, then there's a severely compromised bump-absorbing capacity as the suspension runs out of movement. This can happen at either extension or compression.
One example that many of us are familiar with is fork compression under braking. If the springs are soft enough that the forks are nearly fully compressed under braking, then hitting a bump can use up the tiny bit of remaining travel and seriously upset the motorcycle. It has happened to me; it wasn't fun. The same thing happens when an overloaded vehicle crosses a speed bump; when the suspension is pushed to its limits things can break.
An example of extreme extension at the rear of the bike is climbing a steep rocky hill on a monolever G/S. Under power the suspension is extended much more than normal, so when you ride over a rock the rear wheel cannot extend farther downwards and maintain traction. These are classic examples of BMW's motivation for implementing both the Telelever and the Paralever; each of them is largely unaffected by acceleration and braking, and leaves the suspension in an uncompromised position.
U-joint angles
Some people worry about the two U-joints not being at the same angles, and are convinced that this is a mistake on BMW's part.
The two U-joints in the BMW shaft were never intended to be at the same angle all of the time. Yes, theoretically perfect U-joint cancellation only happens then, but it doesn't happen on our bikes because the suspension designers didn't want it.
The intent of the Paralever is to create a longer effective swingarm (with this length determining the amount of rotation that the final drive experiences as the suspension moves). Making a perfect parallelogram creates a swingarm of infinite length, as far as any shaft-jacking effects go, with no rotation of the rear drive at all. We don't want that because we want a certain amount of jacking to offset the weight changes when we accelerate and decelerate, and we need that rotation to provide it. Putting the effective swingarm pivot up by the center of mass of the bike does this the best.
Rotation of the R100GS Paralever is about half what it is with the K75 Monolever.
So yes, the design is not perfect. But keep in mind that the monolever designs use only one U-joint and therefore have much worse driveline velocity fluctuation. Yes they have rubber dampers, but so does the R100GS and I think the R1100GS, and both of those bikes will probably (haven't done all of the math yet) have less fluctuation than the Monolevers.
This is a separate issue from the ends not being in alignment... as in, not being phased correctly.
Paralever
